Patient-Based Therapeutics
AcuraStem is a patient-based biotechnology company pioneering how treatments are developed for neurodegenerative diseases — including sporadic ALS and FTD — using our proprietary, best-in-class, disease-modeling platform, iNeuroRx®, the gold standard for discovering novel, effective and broadly-acting treatments.
Patient Focused




learn about our work
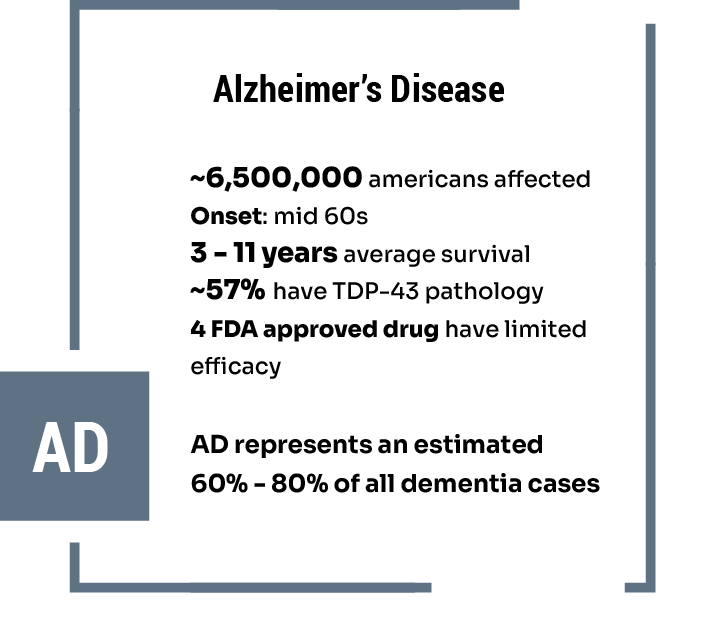
Neurodegenerative Disease Areas


Latest News
Fierce Biotech featured AcuraStem's pioneering work with PIKFYVE inhibitors in a recent article highlighting promising developments in treating both genetic and sporadic forms of ALS. The article discusses two recent studies led by Dr. Justin Ichida's lab at USC and the AcuraStem team that have identified drug candidates with potential to transform the disease course for a larger patient population beyond those with known genetic causes of ALS.
A recent study published in the journal Cell Stem Cell and co-authored by AcuraStem CEO Sam Alworth, M.S., MBA, and AcuraStem co-founder Justin Ichida, Ph.D., et al., shows that suppressing the gene encoding spliceosome-associated factor SYF2 may alleviate symptoms of Amyotrophic Lateral Sclerosis (ALS) and improve neuron survival in both sporadic and genetic forms of the disease.
AcuraStem announced today that CEO Sam Alworth, will present on the company’s latest advances in the development of a PIKFYVE antisense oligonucleotide (ASO) therapeutic for diverse forms of ALS and FTD at the 13th annual California ALS Research Summit
